Here at Isotopx we love serendipity, specifically when an existing instrument can be used for a different application that wasn’t considered when the product was first launched. In an earlier blog I briefly mentioned the use of a stable isotope MS (A.K.A. IRMS) for the measurement of noble gas isotopes. It takes a bit of lateral thinking to understand why you’d do this, and what the gains are, so I thought it was time to take a closer look at this application.

To begin with, some background. I first heard about the use of “pumped” isotope ratio MS for noble gases as opposed to the “static” version only very recently, via some of the team that have published measurements using this technique. Initially I was skeptical, I mean… we sell an instrument that is literally labelled as a noble gas mass spectrometer, so why look elsewhere? The clue is in the sample size. Some readers will be familiar with static mass spectrometers, and why they’re chosen over pumped versions. If you are one of those scientists, feel free to skip the next two paragraphs…
For some noble gas MS measurements, the samples are vanishingly small, potentially coming from exotic sources such as lunar return missions or dust painstakingly collected from the surface of asteroids. These are not to be wasted, every atom counts. The best way to measure these atoms (in the form of ions, obvs) is in the gaseous state, where as many ions as possible can be ingested into the mass spectrometer. Now, for reference, my background is in ICP-MS, a notoriously inefficient way of measuring ions, as most of them are either sucked out of an exhaust or drained into a waste bottle. This is not good for ultra-precious samples. ICP-MS, as with many other mass spectrometric techniques, is a pumped system, whereby the system is continuously introduced to the MS via what I like to think of as a small leak in the vacuum envelope.
So, in the noble gas community it is commonplace to convert the sample to a gas, then inject the entire sample into the mass spectrometer, closing off the inlet. The analysis then takes place, measuring the ions in the gas until most or all of them are consumed. This is a “closed” system, very different from the ICP-MS situation I described above, hence it is given the name static vacuum.
The main benefit of this system is that you are making a very efficient measurement of the available ions. But the downside is that the measurement can only take place for a certain period of time, and over that time period the precision starts to worsen as the remaining sample is depleted.
What about if you have plenty of sample? If you’re collecting entire dewars of gas from the edge of a volcano for example, there is no need to measure every single ion. In this case you might want to consider using a pumped system. Here the benefits and downsides are reversed. Your analysis might take ages, and you’ll consume much more sample, but because the sample is being constantly refreshed, you always have a decent sized ion beam so your precision is improved.

Those readers who have a stable isotope background know what is coming next… the dual inlet. This is a clever device that allows you to automate the alternating introduction of samples and standards. You do this for readily explainable reasons. Lots of measurements can give you great precision, which is great. But, simply taking lots of data points doesn’t necessarily give you great accuracy. If every other measurement is of a known standard, then this data can be used to give you a more accurate measurement of your unknown sample. In other words, the measurements of the known standard are acting as a “reference point” to compare your unknown to.
As an example, if two of your measurements of your standard are reading slightly high, then you know your unknown sample measured between the two is probably reading high by a similar amount. The dual inlet IRMS technique is mature, widely used and widely trusted, and can generate data with excellent accuracy and precision. We are fortunate enough to have access to the field-proven and well liked dual inlet from our sister company Sercon, you can read more about it here.
The obvious next question is how do we apply this to the measurement of noble gas isotopes, and why hasn’t it already been done routinely? The short answer is that several groups have indeed used this technique for the noble gases, but they have faced some tough challenges. The group I am aware of is the team at Woods Hole (WHOI) run by Alan Seltzer (since relocated). I hereby apologize to other groups that have similar used the technique, it’s simply the case that I am aware of the WHOI group through person communication. Here is a link to one of the key papers from the WHOI group.
The team at WHOI used a Thermo MAT 253 IRMS equipped with a dual inlet and they achieved remarkable precision (single figure ppm precision actually!). So, job done? Not quite, as there were a few extenuating factors that made the analysis more difficult, particularly with regard to achieving accuracy. The main factor was the “tail” from 40Ar when measuring 36Ar and 38Ar. Many mass spectrometrists know all about the abundance sensitivity issue; the tail at low mass due to the “smearing” of ion energies. It’s only usually a factor if you are trying to measure a very small ion beam close in mass to a very big ion beam. Unfortunately, that’s exactly what we have for argon, with the 40Ar peak dominating the spectrum.

When measuring the smaller isotopes such as 38Ar, the team at WHOI were able to make a correction for the tail due to the 40Ar. But unfortunately, the error in the correction is large enough to affect the overall accuracy and precision of the measurement, not ideal. The measurement of other noble gas isotope systems can result in other difficulties to overcome, such as having a dynamic range wide enough to include all the necessary isotopes without resorting to ion counting detectors – meaning messy cross-calibrations and a reduction in potential data quality. A great example of this is xenon. The measurement of the smaller isotopes (such as 124Xe and 126Xe) is currently quite tricky to do at any degree of precision.

Can Isotopx help? Hopefully we can. Our SIRIX large radius IRMS was developed for different applications, mainly isotopologues, but lends itself very well to this application. SIRIX uses our ATONA detection system, which means that you can put huge ion beams and tiny ion beams onto the same detectors without any compromises. This is ideal for those isotope systems where there are a broad range of ion beam sizes. And if you want to measure really tiny ion beams then it can also be configured with a mixture of ion counters and Faraday detector. All of the detectors are independently moveable (the collector system is basically the same as Phoenix TIMS) meaning it is easy to change from one isotope system to another.

What about that aforementioned abundance sensitivity issue? We can help there too. Being very closely related to our TIMS instrument, SIRIX has a really rather good abundance sensitivity, utilizing all metal seals, stainless steel construction, and a hefty pumping system. At m/z 38, the contribution from 40Ar is about half a ppm, meaning the measurement of those smaller argon isotopes should be much easier.
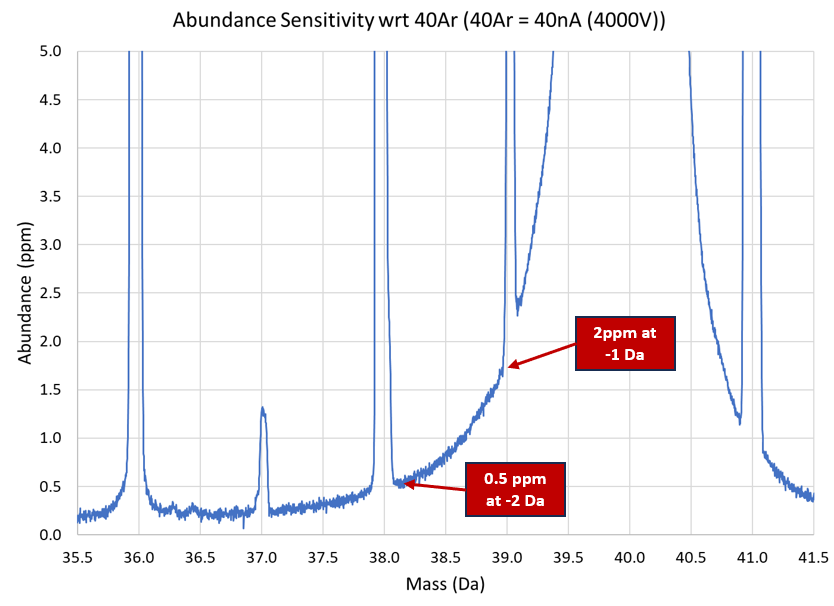
At time of writing we’re taking some argon data, and it looks very promising. Achieving the required precision was not difficult (we can use big beams remember), and achieving the accuracy and longer-term precision is where we are focusing next. We’re doing this using a dual inlet from our sister company Sercon and we expect to be showcasing some excellent data very soon. What this space!
That’s all for now. If you have any comments about how to ease the measurement of noble gas isotopes, especially those at low abundance, please let me know. As always, please send me your thoughts and comments, direct to (Stephen.guilfoyle@isotopx.com). More next time!
